Pigmento rojo 170
| nombre del producto | PIGMENT RED 170 |
| Sinónimos | C.I.Pigment Red 170; C.I.PR170; PR170; P.R.170 |
| CI | 12475 |
| NO CAS. | 2786-76-7 |
| EINECS | 220-509-3 |
| Peso molecular | 454.48 |
| Fórmula molecular | C26H22N4O4 |
| Color | Polvo rojo |
![]()
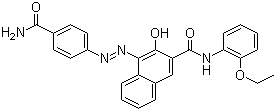
Fórmula de estructura molecular:
Fastness Properties of Pigment Red 170:
| Resistencia a la luz | 5 |
| Resistencia al calor (℃) | 180 |
| Resistencia al agua | 5 |
| Resistencia al aceite | 4 |
| Resistencia ácida | 5 |
| Resistencia a los álcalis | 5 |
| Resistencia al alcohol | 4-5 |
Aplicación principal: Tinta a base de agua, Tinta offset, Tinta a base de solvente, Plástico, Pintura, Impresión textil
Tenemos varios grados y propiedades de pigmentos para satisfacer las necesidades de los diferentes clientes; especifique su aplicación y requisitos para que podamos recomendarlos en consecuencia. Correo electrónico: sy@sypigment.com
Product Description of Pigment Red 170:
Pigment Red 170 has stronger than Pigment Red 210, it is a neutral red color and has two crystal forms; the transparent type is blue-red and has a light fastness of level 6, and the non-transparent type has a light fastness of level 7, high hiding power and more stable to solvents; Novoperm Red F2RK The specific surface area of 70 is 23m2/g; it is mainly used in coatings, and can be mixed with molybdenum chrome orange and quinacridone.Mainly used in coatings and solvent printing inks, water-based printing inks, textile printing and dyeing, etc.
Pigment red 170 or PR170 is an organic pigment extensively used in automotive coatings and painting.
It is produced synthetically by converting p-aminobenzamide into the corresponding diazonium compound followed by coupling with 3-hydroxy-2-naphththoic acid (2-ethoxy)anilide ("Naphtol AS-PH" dye precursor).
In the solid state the hydrazo tautomer forms and several crystal structures exist. In the initial α polymorph the molecules are arranged in a herringbone pattern with extensive hydrogen bonding. The φ polymorph is more dense and more stable and produced industrially by thermal treatment in water at 130°C under pressure. In this phase the molecules are planar and arranged in layers. Extensive hydrogen bonding exists within the layer but between layers the only interactions are Van der Waals forces. Dense crystal structures are preferred for pigments used in coatings because in the event of photochemical decomposition the fragments are locked in place and are able to recombine. Research shows that by replacing the ethoxy group in this compound by a methoxy group the crystal structure is less stable and in the final application and the color fades more easily. By careful selection of substituents it is possible to optimize crystal structure and improve optical properties.
TDS (Pigment Red 170) MSDS (Pigment Red 170)Sinónimos
- C.I. Pigment Red 170
- 2786-76-7
- PIGMENT RED 170
- UNII-54O6PK8790
- 54O6PK8790
- 4-[(4-carbamoylphenyl)diazenyl]-N-(2-ethoxyphenyl)-3-hydroxynaphthalene-2-carboxamide
- 4-[[4-(Aminocarbonyl)phenyl]azo]-N-(2-ethoxyphenyl)-3-hydroxynaphthalene-2-carboxamide
- CI Pigment Red 170
- 2-Naphthalenecarboxamide, 4-((4-(aminocarbonyl)phenyl)azo)-N-(2-ethoxyphenyl)-3-hydroxy-
- 2-Naphthalenecarboxamide, 4-[[4-(aminocarbonyl)phenyl]azo]-N-(2-ethoxyphenyl)-3-hydroxy-
- 4-4-(Aminocarbonyl)phenylazo-N-(2-ethoxyphenyl)-3-hydroxynaphthalene-2-carboxamide
- Permanent Red F 5RK
- Permanent Red F 3RK70
- C.I. Pigment Red 120
- HSDB 3901
- 4-((4-(Aminocarbonyl)phenyl)azo)-N-(2-ethoxyphenyl)-3-hydroxynaphthalene-2-carboxamide
- EINECS 220-509-3
- EC 220-509-3
- SCHEMBL2139449
- DTXSID7029243
- SCHEMBL12954916
- SCHEMBL14560976
- SCHEMBL16191196
- SCHEMBL21468586
- P.R.266F7RK
- 2-Naphtho-o-phenetidide, 4-((p-carbamoylphenyl)azo)-3-hydroxy-
- ZINC33839056
- ZINC100048551
- 4-((4-(Aminocarbonyl)phenyl)azo)-N-(2-ethoxyphenyl)-3-hydroxy-2-naphthalenecarboxamide
- C.I.12475
- 071N997
- W-109137
- Q15425799
- 2-Naphthalene-carboxamide,3-hydroxy-4-(((4-aminocarbonyl)phenyl)azo)-N-(2-ethoxyphenyl)-
- 3-(2-Ethoxyphenylcarbamoyl)-1-[2-(4-carbamoylphenyl)hydrazono]naphthalene-2(1H)-one
- 4-[(E)-(4-carbamoylphenyl)diazenyl]-N-(2-ethoxyphenyl)-3-hydroxy-2-naphthamide
- 2-Naphthalenecarboxamide, 4-(2-(4-(aminocarbonyl)phenyl)diazenyl)-N-(2-ethoxyphenyl)-3-hydroxy-
Nombre IUPAC: 4-[(4-carbamoylphenyl)diazenyl]-N-(2-ethoxyphenyl)-3-hydroxynaphthalene-2-carboxamide
InChI: InChI=1S/C26H22N4O4/c1-2-34-22-10-6-5-9-21(22)28-26(33)20-15-17-7-3-4-8-19(17)23(24(20)31)30-29-18-13-11-16(12-14-18)25(27)32/h3-15,31H,2H2,1H3,(H2,27,32)(H,28,33)
InChIKey: BGVYDWVAGZBEMJ-UHFFFAOYSA-N
SONRISAS canónicas: CCOC1=CC=CC=C1NC(=O)C2=CC3=CC=CC=C3C(=C2O)N=NC4=CC=C(C=C4)C(=O)N
| Nombre de la propiedad | El valor de la propiedad |
| Peso molecular | 454.5 |
| XLogP3-AA | 5.2 |
| Recuento de donantes de enlaces de hidrógeno | 3 |
| Recuento de aceptores de enlaces de hidrógeno | 6 |
| Recuento de bonos giratorios | 7 |
| Masa exacta | 454.1641052 |
| Masa monoisotópica | 454.1641052 |
| Área de superficie polar topológica | 126 Ų |
| Recuento de átomos pesados | 34 |
| Cargo formal | 0 |
| Complejidad | 724 |
| Recuento de átomos de isótopos | 0 |
| Recuento de estereocentros de átomos definidos | 0 |
| Recuento de estereocentros de átomos indefinidos | 0 |
| Recuento de estereocentros de enlaces definidos | 0 |
| Recuento de estereocentros de enlace indefinido | 0 |
| Recuento de unidades unidas covalentemente | 1 |
| El compuesto está canonizado | Sí |



